
Citation: Mahmoud AM and Hussein OE. Anti-Diabetic Effect of Naringin: Insights into the Molecular Mechanism. Diabetes Obes Int J 2016, 1(5): 000128.
*Corresponding author: Ayman M Mahmoud, Physiology Division, Zoology Department, Faculty of Science, Beni-Suef University, Salah Salim St., 62514, Beni-Suef, Egypt. Tel: +201144168280; Email: ayman.mahmoud@science.bsu.edu.eg
Diabetes mellitus is a metabolic disorder characterized by chronic hyperglycemia resulting from either defect in insulin secretion or insulin action [1,2]. In 2015, the International Diabetes Federation (IDF) anticipated that the number of diabetes patients was 415 million and is expected to increase to 642 million by 2040 [3]. If ineffectively controlled, chronic hyperglycemia can cause numerous complications in different body organs [3]. Because of the limitations and side effects of the current therapeutic options for the management of diabetes, the search for safe and effective alternative anti-diabetic agents became a necessity.
Flavonoids are a group of natural polyphenolic compounds with potential benefits in human health [4]. Research from our laboratory showed significant antioxidant, anti-inflammatory, anti-diabetic, cardioprotective and hepatoprotective effects of flavonoids [2,5-10]. Naringin and its aglycone naringenin are two flavonoid compounds with promising anti-diabetic effects. We have previously reported the potent anti-diabetic effects of naringin, both in vivo and in vitro [2,6,9]. In our studies, we have demonstrated the ability of naringin to improve blood glucose levels through its ability to improve insulin sensitivity and secretion, attenuate inflammation and oxidative stress, enhance peripheral glucose uptake, decrease intestinal glucose and cholesterol absorption and suppress hepatic glucose production. In addition, naringin showed marked modulatory effect on adiponectin and resist in levels, adipose tissue peroxisome proliferator activated receptor gamma (PPARγ) expression, and muscle glucose transporter (GLUT)-4 translocation [9,11,12].
In other studies, naringin showed ability to decrease the activity of 3-hydroxy-3-methyl-glutaryl-coenzyme A (HMG-CoA) reductase and thus decrease blood lipids [12- 14]. Naringin has also been reported to regulate the plasma lipids in hypercholesterolemic animals fed a high fat diet [15,16]. In the liver of db/db mice, naringin modified the activities of hepatic lipid-metabolizing enzymes and improved plasma lipid metabolism [16]. In Type 2 diabetic mice, naringin up-regulated hepatic PPARγ and GLUT4, and regulated the expression of hepatic enzymes involved in glycolysis and gluconeogenesis, thereby improving hyperglycemia [16,17]. In hypercholesterolemic individuals, daily consumption of naringin suppressed the biosynthesis of hepatic cholesterol and decreased plasma low-density lipoprotein (LDL)-cholesterol [15]. These findings supporting the notion that naringin may play a vital role in preventing diabetes as well as obesity.
Taken together, naringin is an effective biomolecule that can play a promising role in treating diabetes and its complications. Naringin showed marked modulatory effects on insulin sensitivity and secretion, glucose transporters, PPARγ, hepatic glucose production, peripheral glucose uptake, blood lipids, cholesterol biosynthesis, intestinal glucose absorption, oxidative stress and inflammation (summarized mechanistic pathways are represented in Figure 1). No doubt that there are other beneficial effects to consider; therefore, additional studies are needed to promote our understanding of the exact mechanistic pathways mediating the anti-diabetic effect of naringin.
AbbreviationsAMPK, 5' adenosine monophosphateactivated protein kinase; GLUT, glucose transporter; LPL, lipoprotein lipase; HMG-CoA, 3-hydroxy-3-methylglutaryl-coenzyme A; G6PD, glucose-6-phosphate dehydrogenase; PEPCK, phosphoenolpyruvate carboxykinase; G6Pase, glucose 6-phosphatase; PPARγ, peroxisome proliferator activated receptor gamma; NF- κB, nuclear factor-kappaB; TNF-α, tumor necrosis factor alpha; IL, interleukin; LPO, lipid peroxidation; NO, nitric oxide; GSH, reduced glutathione; SOD, superoxide dismutase; CAT, catalase; GPx, glutathione peroxidase.
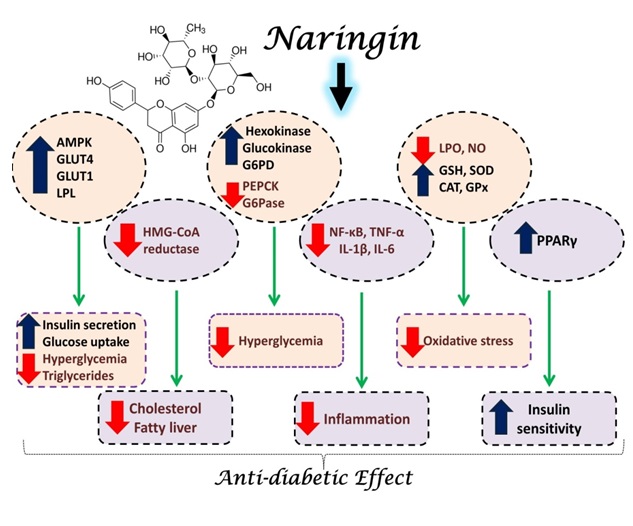
Figure 1: Schematic presentation of the anti-diabetic mechanism of naringin.
Chat with us on WhatsApp