
Citation: Ďurišová M*. Mathematical Model of the Pharmacokinetic Behavior of Warfarin. Adv Pharmacol Clin Trials 2016, 1(1): 000107.
*Corresponding author: Mária Ďurišová, Slovak Academy of Sciences, Institute of Experimental Pharmacology and Toxicology, Bratislava Slovak Republic, Tel: 00421254779928; Email:maria.durisova@savba.sk
The current study is a companion piece of an earlier study by Colburn published in August 1983 Issue of the Journal of Pharmacokinetics and Biopharmaceutics; therefore the data published in the study cited here were used. For modeling purposes, an advanced mathematical modeling method based on the theory of dynamic systems was employed. The modeling method employed, has been introduced to pharmacokinetics in the study by Dedík et al. entitled: “Estimation of influence of gastric emptying on shape of glucose concentration-time profile measured in oral glucose tolerance test”, published in September 2007 Issue of the journal Diabetes Research and Clinical Practice. The goal of the current study was to continue presenting some interesting views on a successful use of an advanced mathematical modeling method based on the theory of dynamic systems in a pharmacokinetic study. The mathematical model of the pharmacokinetic behavior of warfarin was developed using the data from the study cited above. The mathematical model developed successfully described the pharmacokinetic behavior warfarin in the subject described in the study by Colburn.
Keywords: Intravenous; Administration; Dynamic system; Mathematical model
Warfarin is an essential anticoagulant with a narrow therapeutic index. It is one of widely prescribed anticoagulants such as: aspirin, dabigatran, rivaroxaban, apixaparin, lovenox, fondaparinux and heparin. Commercially available warfarin is a racemic mixture of two enantiomers that are extensively metabolized in liver. Warfarin is commonly used for prophylaxis and treatment of venous and arterial thrombosis [1-11].
The current study is a companion piece of the related study by Colburn published in August 1973 Issue of the Journal Pharmacokinetics and Bio pharmaceutics. Therefore, the data from the study cited here were used [1]. For modeling purposes, an advanced mathematical modeling method based on the theory of dynamic systems was employed, see for example the following studies [12- 27] and references therein. The goal of the current study was to continue presenting some interesting views on a successful use of an advanced modeling method based on the theory of dynamic systems in pharmacokinetics.
Previous examples showing how an advanced modeling method used in the current study can be employed in various pharmacokinetic studies can found in the full text articles, authored and/or co-authored by the author of the current study, which can be downloaded free of charge from the following web page of the author: http://www.uef.sav.sk/advanced.htm.
MethodsThe data from the study by Colburn [1] were used. As stated above, an advanced mathematical modeling method based on the theory of dynamic systems [12-27] was employed for modeling purposes. Throughout the current study, the lower case letter “S” denoted the complex Laplace variable. The development of a mathematical model of the dynamic system H was performed in the following successive steps:
First, the dynamic system, denoted by H was defined using: 1) the Laplace transform of the mathematically approximated the subject’s plasma concentration-time profile of warfarin after the intravenous administration of warfarin, denoted by C(s), and considered the output of the pharmacokinetic dynamic system H and 2) the Laplace transform of the mathematically approximated the intravenous administration of warfarin to a subject [1], denoted by I(s), and considered the warfarin input to the subject’ body and/or to the dynamic system H [13- 28]. In the following text, the pharmacokinetic dynamic system H, was simply called the dynamic system.
Second, the following simplifying assumptions were made: a) initial conditions of the dynamic system H were zero; b) pharmacokinetic processes occurring in the subject’s body after the intravenous administration of warfarin were linear and time invariant [13-27]; c) concentrations of warfarin were the same throughout all subsystems of the dynamic system (where a subsystem was an integral part of the dynamic system H); d) no barriers to the distribution and /or elimination of warfarin existed; e) the dynamic system H was stable them and did not become unstable during the time course of the study published previously [1] and the current study.
Third, the dynamic system H was used to mathematically describe static and dynamic properties [28-31] of the pharmacokinetic behavior of warfarin in a subject [1]. Fourth, the transfer function, denoted by H(s) of a the dynamic system was derived using: 1) the Laplace transform of the mathematically approximated plasma concentration-time profile of warfarin of a subject, denoted by C(s), and considered the output of the dynamic system H, and 2) the Laplace transform of the mathematically approximated intravenous administration of warfarin to a subject and considered warfarin input I(s), to the subject’s body and/or to the dynamic system H [12-28], see the following equation:
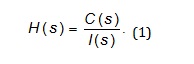
Fifth, the dynamic system H was approximated with the transfer function, denoted by H(s).
Sixth, the computer program named CTDB [13] and the transfer function model HM(s) approximated by Eq. (2), were used to develop a mathematical model of the dynamic system H [12-27].
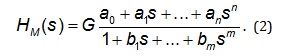
On the right-hand-side of Eq. (2) is the Padé
approximant [29,30] of the transfer function model HM(s)
G is an estimator of the model parameter called a gain of a
dynamic system, a1… an, b1… bm are additional model
parameters, n is the highest degree of the nominator
polynomial, and m is the highest degree of the
denominator polynomial, where n Seventh, the transfer function H(s) was converted into
the equivalent frequency response function, denoted by
F(iωj) [13-26]. Eighths, the non-iterative method published
previously [29] was used to develop a mathematical
model of frequency response function, FM(iωj) described
by Equation 3, and to determine point estimates of
parameters of the frequency response function model FM
(iωj) in the frequency domain [13-26].
Analogously as in Eq. (2), n is the highest degree of the
numerator polynomial of the frequency response function
model FM(iωj) m is the highest degree of the denominator
polynomial of the frequency response function model
FM(iωj) where n≤m, i is the imaginary unit, and ω is the
angular frequency in Eq. (3). Ninth, the Akaike information criterion (AIC), modified
for the use in the complex domain [14] was used to select
the best model of the frequency response function FM(iωj)
and to determine point estimates of the parameters of the
best model of the frequency response function FM(iωj) in
the complex domain. After that, the Monte-Carlo and the
Gauss-Newton method [31-33] were used to refine the
best model of the frequency response function FM(iωj) and
to determine 95 % confidence intervals of the parameters
of the model of the frequency response function FM(iωj) in
the time domain. After the development of a mathematical model of the
dynamic system H, the following potentially important
pharmacokinetic variables were determined: the
elimination half-time of warfarin, denoted by t1/2 , the
area under the plasma concentration-time profile of
warfarin from time zero to infinity, denoted by AUC0-∞, the
total body clearance of warfarin, denoted by Cl. The
maximum serum concentration of warfarin, denoted by
Cmax, was read directly from the plasma concentrationtime
profile of warfarin of the subject. The best-fit third-order model, FM(iωj) selected using
AIC, is described by the following equation:
This third-order model provided an adequate fit to the
warfarin plasma concentration-time profile of warfarin
[1] Estimates of the model parameters G, a0, a1, b1, b2, b3
are in Table 1. Model-based estimates of potentially important pharmacokinetic variables of warfarin are in Table 2. Figure 1 illustrates the observed plasma concentration
time profile of warfarin and the description of the
observed profile with the developed model of the
dynamic system which mathematically approximated
dynamic and static properties of the pharmacokinetic
behavior of warfarin in the subject investigated [1].
Model-based estimates of potentially important
pharmacokinetic variables of warfarin are listed in (Table
2). The most general form of a model of a frequency
response function FM(iωj), which was used in the current
study is described by Eq. (4). The transfer function model
HM(s) and the frequency response function model
FM(iωj) are implemented in the computer program CTDB
[13]. A demo version of the computer program CTDB is
available at: http://www.uef.sav.sk/advanced.htm. The dynamic system used in the current study was a
mathematical object, without any physiological
significance. It was used to mathematically approximate
static and dynamic properties of the pharmacokinetic
behavior of warfarin in the subject investigated [1,34-36].
The advanced modeling method used in the current study
has been described in detail in the previous studies
authored and/or co authored by the author of the current
study [13-26], therefore the model description has not
been given here. Analogously as in the studies published previously [13-
26], the development of a mathematical model the
dynamic system was based on the known input and
output of the dynamic system under study, in the current
study. In general, if a dynamic system is modeled using a
transfer function model; as it was the case in the current
study (see Eq. (2)), then the accuracy of the model
depends on the degrees of the polynomials of the transfer
function model used to fit the data, see for example the
following studies [13-26] and references therein. The parameter gain is called also a gain coefficient,
and/or a gain factor. In general, a parameter gain is
defined as a relationship between a magnitude of an
output of a dynamic system output to a magnitude of a
dynamic system input in steady state. Or in other words, a
parameter gain of a dynamic system is a proportional
value that shows a relationship between a magnitude of a
dynamic system output to a magnitude of a dynamic
system input in steady state. The pharmacokinetic
meaning of a parameter gain depends on the nature of the
dynamic system under study; see for example full text
articles available free of charge at:
http://www.uef.sav.sk/advanced.htm. The non-iterative
modeling method described in the study published
previously [29] and used in the current study enables
quick identification of an optimal structure of a model of a
frequency response. It is a great advantage of this method,
because it significantly speeds up the development of
frequency response models. The reason for conversion of HM(s) to FM(iωj) has been
explained in the studies published previously; see for
example the studies [20-26], therefore an explanation
was not given in the current study. The mathematical
model developed in the current study sufficiently
described static and dynamic properties [34-36] of the
pharmacokinetic behavior of warfarin in the subject
investigated [1]. The current study showed again that a
mathematical modeling method based on the theory of
dynamic systems can be successfully used in
mathematical modeling in pharmacokinetics. Frequency response functions are complex functions,
therefore modeling must be performed in the complex
domain. In general, modeling methods used to develop
mathematical models of frequency response functions are
computationally intensive. Furthermore, the methods
considered require at least a partial knowledge of the
theory of dynamic system, and an abstract way of thinking
about a dynamic system under study. The principal difference between traditional
pharmacokinetic modeling methods and modeling
methods that use of modeling and computational tools
from the theory of dynamic systems can be explained as
follows: the former methods are based on mathematical
modeling plasma and/or blood concentration-time
profiles of drugs administered, however the latter
methods are based on mathematical modeling dynamic
relationships between a mathematically described a drug
administration and a mathematically described resulting
plasma (or/or blood) concentration-time profiles of drugs
administered. See, for example, the full text articles and
an explanatory picture, available free of charge at the
following web page of the author:
http://www.uef.sav.sk/advanced.htm. The computational and modeling methods that use
computational and modeling tools from the theory of
dynamic systems can be used for example for adjustment
of a drug (or a substance) dosing, aimed at achieving and
then maintaining required drug (or a substance)
concentration–time profile in a patient see the following
study [17]. Moreover, the methods considered here can
be used for safe and cost-effective individualization of
dosing of a drug or a substance, for example using
computer-controlled infusion pumps [37,38]. This is very
important for example for an administration of a clotting
factor to a hemophilia patient, as exemplified in the
simulation study [17]. The advantages of the model and modeling method
used in the current study are evident here: The models
developed overcome the well known limitations of
compartmental models: For the development and use of
the models considered here, an assumption of well-mixed
spaces in the body (in principle unrealistic) is not
necessary. The basic structure of the models is broadly
applicable. Therefore, this structure can be used in the
development of mathematical models not only in the field
of pharmacokinetics but also in several other scientific
and practical fields. From a point of view of the
pharmacokinetic community, an advantage of the models
developed using computational tools from the theory of
dynamic systems is that the models considered here
emphasize dynamical aspects of the pharmacokinetic
behavior of an administered drug in a human and/or in an
animal body. Transfer functions of dynamic systems are
not unknown in pharmacokinetics; see for example the
following studies [37,38]. In pharmacokinetics, transfer
functions are usually called disposition functions [39,40].
An anticoagulant therapy is a medication that affects the
blood clotting process. Therefore anticoagulants and the
knowledge of pharmacokinetics of anticoagulants are
important for medical practice. The author assumes that
the current study may contribute to the knowledge base
in pharmacokinetics. The model developed and used in the current study
successfully described the pharmacokinetic behavior of
warfarin in the subject investigated [1] after the
intravenous administration warfarin. The modeling
method used in the current study is universal; therefore it
is applicable to any kind of a dynamic system, not only in
the field of pharmacokinetics but also in many other
scientific or practical fields. The current study again
showed that a mathematical modeling method based on
the theory of dynamic systems can be advantageously
used in pharmacokinetic modeling. To see the previous
examples illustrating the successful use of the modeling
method employed in the current study please visits the
author’s web page (an English version):
http://www.uef.sav.sk/advanced.htm. The current study
reaffirmed that an integration of key concepts from
pharmacokinetic and bioengineering is a good and
efficient way to study dynamic processes in
pharmacokinetics, because such integration combines
mathematical rigor with biological insight. There is no conflict of interest. The author gratefully acknowledges the financial
support obtained from the Slovak Academy of Sciences in
Bratislava, Slovak Republic.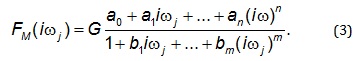
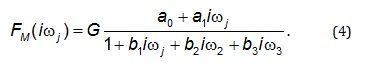
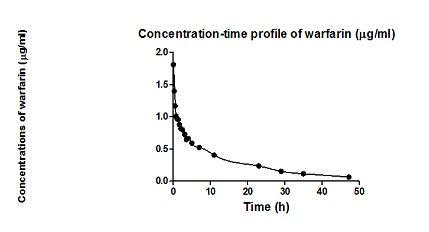
Figure 1: Observed plasma concentration time profile of warfarin and the description of the observed profile with the developed model of the dynamic system, mathematically describing the static and dynamic properties of the pharmacokinetic behavior of warfarin the subject investigated [1].
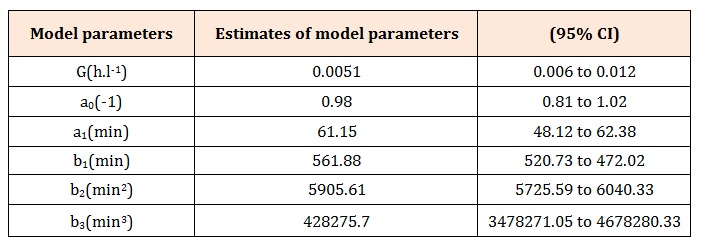
Table 1: Parameters of the third-order model of the dynamic system describing the pharmacokinetic behavior of intravenously administered warfarin in the subject investigated [1].
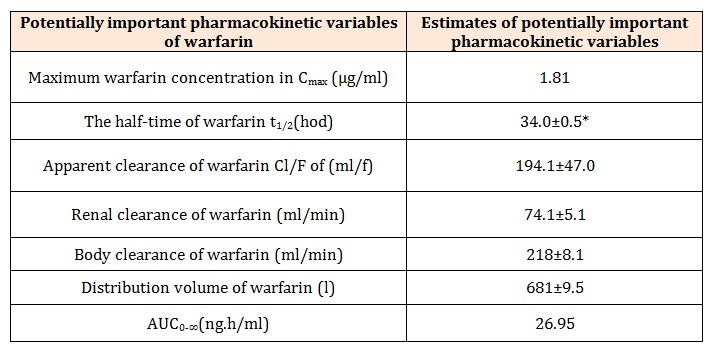
Table 2: Model-based estimates of potentially important pharmacokinetic variables of intravenously administered warfarin of the subject investiged [1].
*standard deviation